2020 Latest Design Apo Hydrochlorothiazide - Pitavastatin Calcium – CPF
2020 Latest Design Apo Hydrochlorothiazide - Pitavastatin Calcium – CPF Detail:
| 匹伐他汀钙 | Pitavastatin | 147526-32-7 | In-House |
| PI-5 | 3800-06-4 | In-House | |
| PI-6 | 145516-11-4 | In-House | |
| PI-7 | 121660-11-5 | In-House |
Description
Pitavastatin Calcium (NK-104 hemicalcium) is a potent hydroxymethylglutaryl-CoA (HMG-CoA) reductase inhibitor. Pitavastatin Calcium (NK-104 hemicalcium) inhibits cholesterol synthesis from acetic acid with an IC50 of 5.8 nM in HepG2 cells. Pitavastatin Calcium is an efficient hepatocyte low-density lipoprotein-cholesterol (LDL-C) receptor inducer. Anti-cancer activity.
Background
Pitavastatin Calcium is a competitive inhibitor of the enzyme HMGCR (HMG-CoA reductase) results in a reduction in LDL cholesterol synthesis. Alternate studies show that pitavastatin can suppress oxygen production in endothelial cells by inhibiting NADPH oxidase. In addition, pitavastatin reduces the expression of eNOS mRNA while increasing the NO dependent response stimulated by acetylcholine and the calcium ionophore, A23187. Furthermore, pitavastatin inhibits the up-regulation of conductance calcium-activated potassium channels by lowering cholesterol levels in cells.
Storage
4°C, protect from light
*In solvent : -80°C, 6 months; -20°C, 1 month (protect from light)
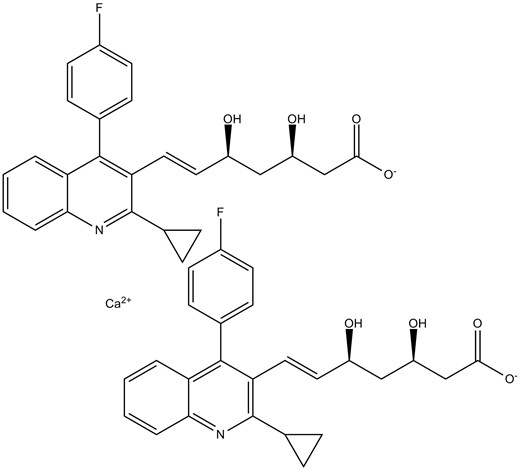
Chemical structure
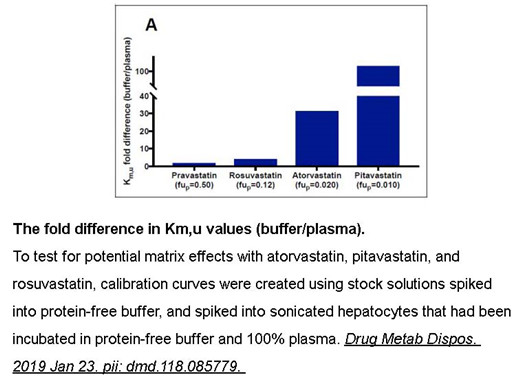
Related Biological Data
Proposal 18 Quality Consistency Evaluation projects which have approved 4, and 6 projects are under approving.

Advanced international quality management system has laid solid foundation for sales.

Quality supervision runs through the whole life cycle of the product to ensure the quality and therapeutic effect.

Professional Regulatory Affairs team supports the quality demands during the application and registration.
VIEW MORE
VIEW MORE
International cooperation

Domestic cooperation

Product detail pictures:
Related Product Guide:
We support our purchasers with ideal high-quality merchandise and significant level company. Becoming the specialist manufacturer in this sector, we now have received loaded practical encounter in producing and managing for 2020 Latest Design Apo Hydrochlorothiazide - Pitavastatin Calcium – CPF , The product will supply to all over the world, such as: Poland, Mauritius, Jamaica, Our Company policy is "quality first, to be better and stronger, sustainable development" . Our pursuit goals is "for society, customers, employees, partners and enterprises to seek reasonable benefit". We aspirate to do cooperate with all different the auto parts manufacturers, repair shop, auto peer , then create a beautiful future! Thank you for taking time to browse our website and we would welcome any suggestions you may have that can help us to improve our site.
As an international trading company, we have numerous partners, but about your company, I just want to say, you are really good, wide range, good quality, reasonable prices, warm and thoughtful service, advanced technology and equipment and workers have professional training, feedback and product update is timely, in short, this is a very pleasant cooperation, and we look forward to the next cooperation!











