OEM Manufacturer Favipiravir - Baloxavir Marboxil 1985606-14-1 – CPF
OEM Manufacturer Favipiravir - Baloxavir Marboxil 1985606-14-1 – CPF Detail:
Background
Baloxavir marboxil is an antiviral drug and is an endonuclease inhibitor. In rats and monkeys, the blood concentration of the drug is lower than the minimum detection amount in a single oral administration of the drug. In preclinical influenza A and B infection models (including strains that are resistant to existing antiviral drugs), Baloxavir marboxil has a certain effect.
Chemical Properties
| Storage | Store at -20°C |
| M.Wt | 571.55 |
| Cas No. | 1985606-14-1 |
| Formula | C27H23F2N3O7S |
| Synonyms | S-033188 |
| Solubility | Soluble in DMSO |
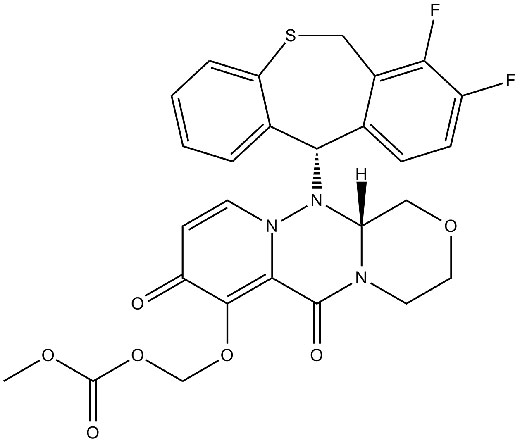
| Chemical Name | (((R)-12-((S)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12a-hexahydro-1H-[1,4]oxazino[3,4-c]pyrido[2,1-f][1,2,4]triazin-7-yl)oxy)methyl methyl carbonate |
| SDF | Download SDF |
| Canonical SMILES | O=C1C=CN(N([C@]2(N(C3=O)CCOC2)[H])[C@H]4C5=CC=C(C(F)=C5CSC6=CC=CC=C46)F)C3=C1OCOC(OC)=O |
| Shipping Condition | Evaluation sample solution: ship with blue ice. All other available sizes: ship with RT, or blue ice upon request. |
| General tips | For obtaining a higher solubility, please warm the tube at 37°C and shake it in the ultrasonic bath for a while. Stock solution can be stored below -20°C for several months. |
Chemical structure
Proposal 18 Quality Consistency Evaluation projects which have approved 4, and 6 projects are under approving.

Advanced international quality management system has laid solid foundation for sales.

Quality supervision runs through the whole life cycle of the product to ensure the quality and therapeutic effect.

Professional Regulatory Affairs team supports the quality demands during the application and registration.
VIEW MORE
VIEW MORE
International cooperation

Domestic cooperation

Product detail pictures:

Related Product Guide:
We strive for excellence, service the customers", hopes to become the best cooperation team and dominator enterprise for personnel, suppliers and customers, realizes value share and continuous promotion for OEM Manufacturer Favipiravir - Baloxavir Marboxil 1985606-14-1 – CPF , The product will supply to all over the world, such as: UAE, Libya, United Arab emirates, With many years good service and development, we've a professional international trade sales team. Our products have exported to North America, Europe, Japan, Korea, Australia, New Zealand, Russia and other countries. Looking forward to build up a good and long term cooperation with you in coming future!
It is not easy to find such a professional and responsible provider in today's time. Hope that we can maintain long-term cooperation.











