Well-designed Thalidomide Story - Apixaban – CPF
Well-designed Thalidomide Story - Apixaban – CPF Detail:
Background
Apixaban is a highly selective and reversible inhibitor of Factor Xa with Ki values of 0.08 nM and 0.17 nM in human and rabbit, respectively[1].
Factor X, also known by the eponym Stuart–Prower factor, is an enzyme of the coagulation cascade. Factor X is activated, by hydrolysis, into factor Xa by both factor IX. Factor Xa is the activated form of the coagulation factorthrombokinase.Inhibiting Factor Xa could offer an alternate method for anticoagulation. Direct Xa inhibitors are popular anticoagulants [2].
In vitro: Apixabanhas exhibited a high degree of potency, selectivity, and efficacy on Factor Xa with Ki of 0.08 nM and 0.17 nM for Human Factor Xa and Rabbit Factor Xa, respectively [1]. Apixaban prolonged the clotting times of normal human plasma with the concentrations (EC2x) of 3.6, 0.37, 7.4 and 0.4 μM, which are required respectively to double the prothrombin time (PT), modified prothrombin time (mPT), activated partial thromboplastin time (APTT) and HepTest. Besides, Apixaban showed the highest potency in human and rabbit plasma, but less potency in rat and dog plasma in both the PT and APTT assays [3].
In vivo: Apixaban exihibited the excellent pharmacokinetics with very low clearance (Cl: 0.02 L kg-1h-1), and low volume of distribution (Vdss: 0.2 L/kg) in the dog. Besides, Apixaban also showed a moderate half-life with T1/2 of 5.8 hours and good oral bioavailability (F: 58%) [1]. In the arteriovenous-shunt thrombosis (AVST), venous thrombosis (VT) and electrically mediated carotid arterial thrombosis (ECAT) rabbit models, Apixaban produced antithrombotic effects with EC50 of 270 nM, 110 nM and 70 nM in a dose-dependent manner[3]. Apixaban significantly inhibited factor Xa activity with an IC50 of 0.22 μM in rabbit ex vivo[4]. In chimpanzee, Apixaban also showed small volume of distribution (Vdss: 0.17 L kg-1), low systemic clearance (Cl: 0.018 L kg-1h-1), and good oral bioavailability (F: 59%) [5].
References:
Pinto D J P, Orwat M J, Koch S, et al. Discovery of 1-(4-methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl) phenyl)-4, 5, 6, 7-tetrahydro-1 H-pyrazolo [3, 4-c] pyridine-3-carboxamide (Apixaban, BMS-562247), a highly potent, selective, efficacious, and orally bioavailable inhibitor of blood coagulation factor Xa[J]. Journal of medicinal chemistry, 2007, 50(22): 5339-5356.
Sidhu P S. Direct Factor Xa Inhibitors as Anticoagulants[J].
Wong P C, Crain E J, Xin B, et al. Apixaban, an oral, direct and highly selective factor Xa inhibitor: in vitro, antithrombotic and antihemostaticstudies[J]. Journal of Thrombosis and Haemostasis, 2008, 6(5): 820-829.
Zhang D, He K, Raghavan N, et al. Metabolism, pharmacokinetics and pharmacodynamics of the factor Xa inhibitor apixaban in rabbits[J]. Journal of thrombosis and thrombolysis, 2010, 29(1): 70-80.
He K, Luettgen J M, Zhang D, et al. Preclinical pharmacokinetics and pharmacodynamics of apixaban, a potent and selective factor Xa inhibitor[J]. European journal of drug metabolism and pharmacokinetics, 2011, 36(3): 129-139.
Apixaban is a highly selective and reversible inhibitor of Factor Xa with Ki values of 0.08 nM and 0.17 nM in human and rabbit, respectively[1].
Factor X, also known by the eponym Stuart–Prower factor, is an enzyme of the coagulation cascade. Factor X is activated, by hydrolysis, into factor Xa by both factor IX. Factor Xa is the activated form of the coagulation factorthrombokinase.Inhibiting Factor Xa could offer an alternate method for anticoagulation. Direct Xa inhibitors are popular anticoagulants [2].
In vitro: Apixabanhas exhibited a high degree of potency, selectivity, and efficacy on Factor Xa with Ki of 0.08 nM and 0.17 nM for Human Factor Xa and Rabbit Factor Xa, respectively [1]. Apixaban prolonged the clotting times of normal human plasma with the concentrations (EC2x) of 3.6, 0.37, 7.4 and 0.4 μM, which are required respectively to double the prothrombin time (PT), modified prothrombin time (mPT), activated partial thromboplastin time (APTT) and HepTest. Besides, Apixaban showed the highest potency in human and rabbit plasma, but less potency in rat and dog plasma in both the PT and APTT assays [3].
In vivo: Apixaban exihibited the excellent pharmacokinetics with very low clearance (Cl: 0.02 L kg-1h-1), and low volume of distribution (Vdss: 0.2 L/kg) in the dog. Besides, Apixaban also showed a moderate half-life with T1/2 of 5.8 hours and good oral bioavailability (F: 58%) [1]. In the arteriovenous-shunt thrombosis (AVST), venous thrombosis (VT) and electrically mediated carotid arterial thrombosis (ECAT) rabbit models, Apixaban produced antithrombotic effects with EC50 of 270 nM, 110 nM and 70 nM in a dose-dependent manner[3]. Apixaban significantly inhibited factor Xa activity with an IC50 of 0.22 μM in rabbit ex vivo[4]. In chimpanzee, Apixaban also showed small volume of distribution (Vdss: 0.17 L kg-1), low systemic clearance (Cl: 0.018 L kg-1h-1), and good oral bioavailability (F: 59%) [5].
References:
Pinto D J P, Orwat M J, Koch S, et al. Discovery of 1-(4-methoxyphenyl)-7-oxo-6-(4-(2-oxopiperidin-1-yl) phenyl)-4, 5, 6, 7-tetrahydro-1 H-pyrazolo [3, 4-c] pyridine-3-carboxamide (Apixaban, BMS-562247), a highly potent, selective, efficacious, and orally bioavailable inhibitor of blood coagulation factor Xa[J]. Journal of medicinal chemistry, 2007, 50(22): 5339-5356.
Sidhu P S. Direct Factor Xa Inhibitors as Anticoagulants[J].
Wong P C, Crain E J, Xin B, et al. Apixaban, an oral, direct and highly selective factor Xa inhibitor: in vitro, antithrombotic and antihemostaticstudies[J]. Journal of Thrombosis and Haemostasis, 2008, 6(5): 820-829.
Zhang D, He K, Raghavan N, et al. Metabolism, pharmacokinetics and pharmacodynamics of the factor Xa inhibitor apixaban in rabbits[J]. Journal of thrombosis and thrombolysis, 2010, 29(1): 70-80.
He K, Luettgen J M, Zhang D, et al. Preclinical pharmacokinetics and pharmacodynamics of apixaban, a potent and selective factor Xa inhibitor[J]. European journal of drug metabolism and pharmacokinetics, 2011, 36(3): 129-139.
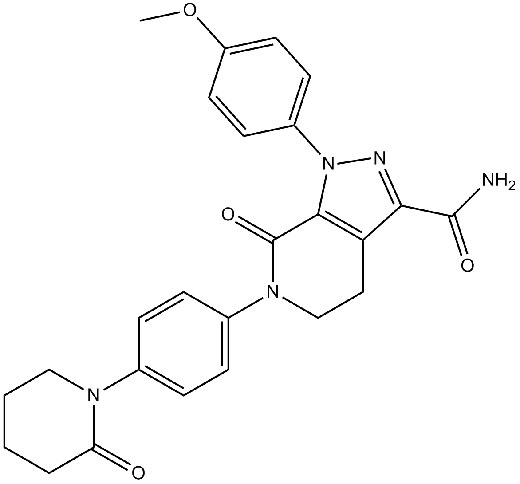
Chemical structure
Proposal 18 Quality Consistency Evaluation projects which have approved 4, and 6 projects are under approving.

Advanced international quality management system has laid solid foundation for sales.

Quality supervision runs through the whole life cycle of the product to ensure the quality and therapeutic effect.

Professional Regulatory Affairs team supports the quality demands during the application and registration.
VIEW MORE
VIEW MORE
International cooperation

Domestic cooperation

Product detail pictures:

Related Product Guide:
We've been commitment to offer the competitive rate ,outstanding merchandise good quality, too as fast delivery for Well-designed Thalidomide Story - Apixaban – CPF , The product will supply to all over the world, such as: Nigeria, India, French, we always keep our credit and mutual benefit to our client, insist our high quality service to moving our clients. always welcome the our friends and clients to come and visit our company and guid our business, if you are interested in our products, you can also submit your purchase information online, and we will contact you immediately, we keep our highly sincere cooperation and wish everything in your side are all well.
This company has a lot of ready-made options to choose and also could custom new program according to our demand, which is very nice to meet our needs.










